|
|
||
| | Gateway | Science & Technology | Chat | Search | News | | ||
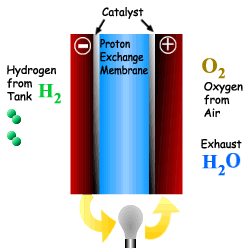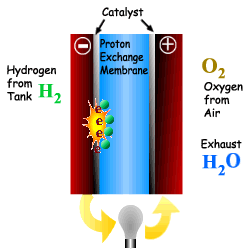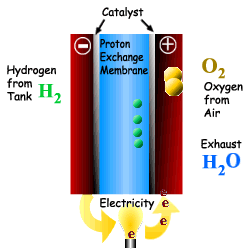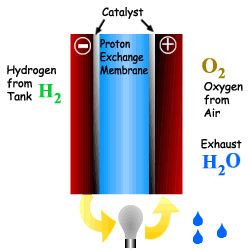
What is a Fuel Cell?
A Fuel Cell is a device that converts chemical
Energy (Potential Energy) directly into electricity via a modified oxidation
process. The process also produces heat, water and possibly CO2
depending upon the fuel used. A Fuel Cell is a device that converts chemical Energy (Potential Energy) directly into electricity via a modified oxidation process. The process also produces heat, water and possibly CO2 depending upon the fuel used. A fuel cell is comprised of an electrolyte, surrounded by two electrodes. The underlying procedure is that oxygen and hydrogen pass over a distinct electrode, thus creating electricity. |
|
||||||||||||||||||||||||||||||
Advantages of Fuel Cells Fuel cells have a number of advantages over conventional power generating equipment: · High efficiency · Low chemical, acoustic, and thermal emissions · Flexibility · Reliability · Low maintenance · Excellent part-load performance · Modularity · Fuel flexibility |
|||||||||||||||||||||||||||||||